Looking for a quick STEM activity that you can do in 20 minutes or less?
This is your answer! You likely already have everything you need to do the Magic Milk science experiment at home right now. Both the prep & cleanup for this experiment is simple.
But being “simple” doesn’t mean it isn’t fun! This awesome experiment is sure to amaze! I was honestly surprised by how well it worked. The results were actually amazing! But first, let’s go over everything you need.
If you’d like more STEM activity ideas, make sure to grab my FREE eBook on 25 STEM activities here.
Table of Contents
Supplies
- 2 % or Whole Milk (no skim milk!)
- Food Coloring
- Cotton Swap
- Dish Soap
How to do the Magic Milk Science Experiment
When I say this is an easy one to do on a whim, I mean it! All you need to do is follow these 4 simple steps.
Step 1: Pour Milk into a Shallow Dish

Pour the milk into a small bowl. Remember, it has to be 2% or whole milk. Skim milk won’t. However, a great way to test hypotheses would be to compare the effects on milk with different fat contents.
Step 2: Add 4-5 Drops of Food Coloring
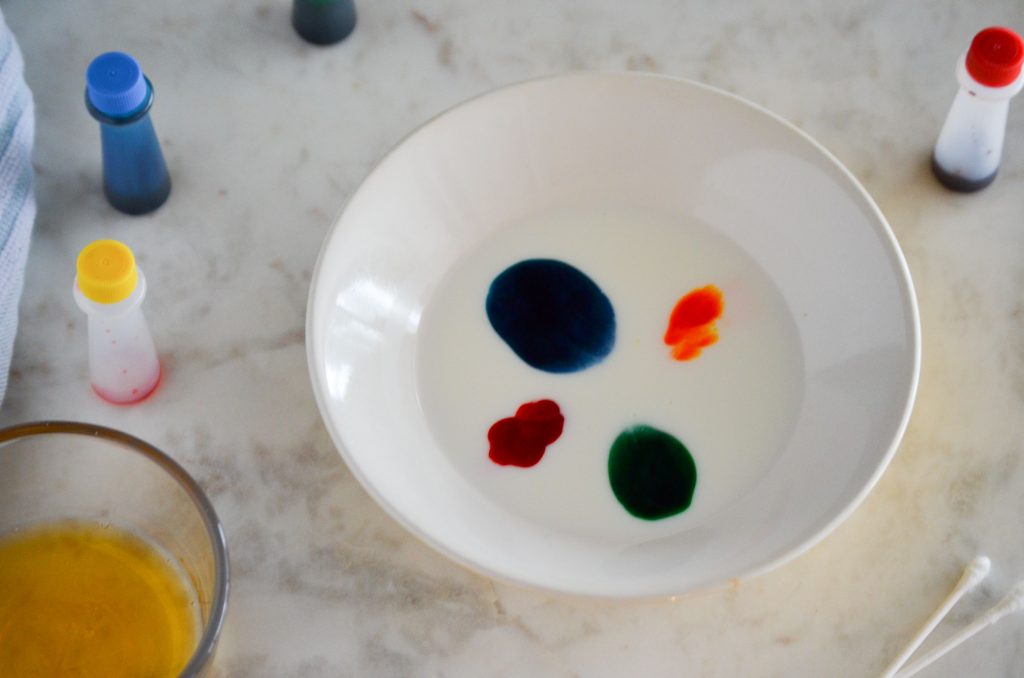
Add a few drops of the food coloring to the milk.
Below we’ll learn how the science experiment works. The food coloring simply allows us to see the effect adding the dish soap to the milk has. The food coloring itself isn’t part of the reaction.
Step 3: Dip Cotton Swap in Dish Soap
Soak the end of your cotton swap with any kind of dish soap.
Step 4: Touch Soapy Cotton Swap to Milk
Be prepared to be amazed!

As soon as you touch the soap to the milk, you’ll see the different colored milk shoot apart.
The reaction is quick, so make sure everyone is watching!
Record what you saw happened. For extra testing, see how the effects are different depending on the type of milk you use: whole, 2% and skim.
How the Magic Milk Science Experiment Works
The secret to the dancing motion of the food coloring is all in the dish soap.
Milk is mostly made up of water, but it also contains vitamins, minerals, and fat. The fat in milk (like all fats) is an example of a nonpolar molecule, which means it does not dissolve in water.
When the soap is added to the milk, the molecules dance around to try to connect to the fat molecules in the milk.
In simplest terms, the soap causes the fat in the milk to separate from the water, which is why this won’t work with skim milk.
So why the food coloring? Without the food coloring, we wouldn’t be able to see how the milk fat molecules rapidly move around once the soap is added. Plus, its an awesome firework effect!







